The word “Rubber” refers to the material derived from the rubber tree originally; however, it refers to the material having large deformability and certain resilience after elimination of the deformation force in now days. Currently, those synthetic materials such as synthetic rubber, fluorinated silicone rubber, silicone rubber, EPDM and natural rubber belong to the same homdogy. In rubber chemistry industry, “synthetic” natural rubber has been developed, which can copy the chemical structure and characteristics of such natural product.
Rubber is the material meeting the demand of many projects, which can be used to ensure the vibration, noise, oscillation and corrosion resistance, abrasiveness, electrical and heat insulation and watertightness, and can also be used in other constraining materials and bearings. The extensibility of rubber can reach more than 100%. Although it is easy to deform under external force, it will return to its original form immediately after the external force is removed, from which we can obviously see the properties of macromolecular substance.
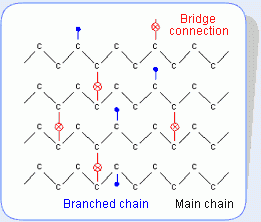
The following figure shows the chemical construction of plastics also classified as macromolecular. Both the rubber and plastic are made of the macromolecular chain (main chain) repeatedly bonded with smallest carbon-carbon unit. Compared with the plastic, in the construction of macromolecule rubber, the sub-chains and macromolecule chains are not bonded together, the macromolecules are cross-linked together via bridge connection, and the so-called elastic property is also attributed to the bridge connection and the branched chain of macromolecule main chain. Such bridging construction makes FFKM very stable under high temperature and pressure conditions.


